Phase Volume Correlations
With Piper, you can calculate in-situ phases as a function of temperature and pressure by way of either the Wilson equilibrium ratio method or the Peng Robinson equation of state.
Piper includes the Wilson equilibrium ratio calculation. This is the most basic equilibrium ratio method and is also used as a first iteration approximation of equilibrium ratios for the Peng-Robinson equation of state.
The Wilson equilibrium ratio correlation is as follows:
![]()
Where ![]() is calculated for each component
and where
is calculated for each component
and where ![]() are all stored parameters for
each component.
are all stored parameters for
each component.
Peng-Robinson Equation of State
Piper uses the Peng Robinson equation of state to solve for phase volumes in the pipeline. Piper requires a 12 component compositional definition to use Peng Robinson.
Piper currently does not perform volume translation on its Peng-Robinson solution.
Water is also currently considered outside of the equation of state model.
The following iterative process is used to calculate equilibrium ratios and from that the molar volumes of the gas and oil phases. Note that Wilson equilibrium ratios are calculated as the first guesses of the equilibrium ratios.
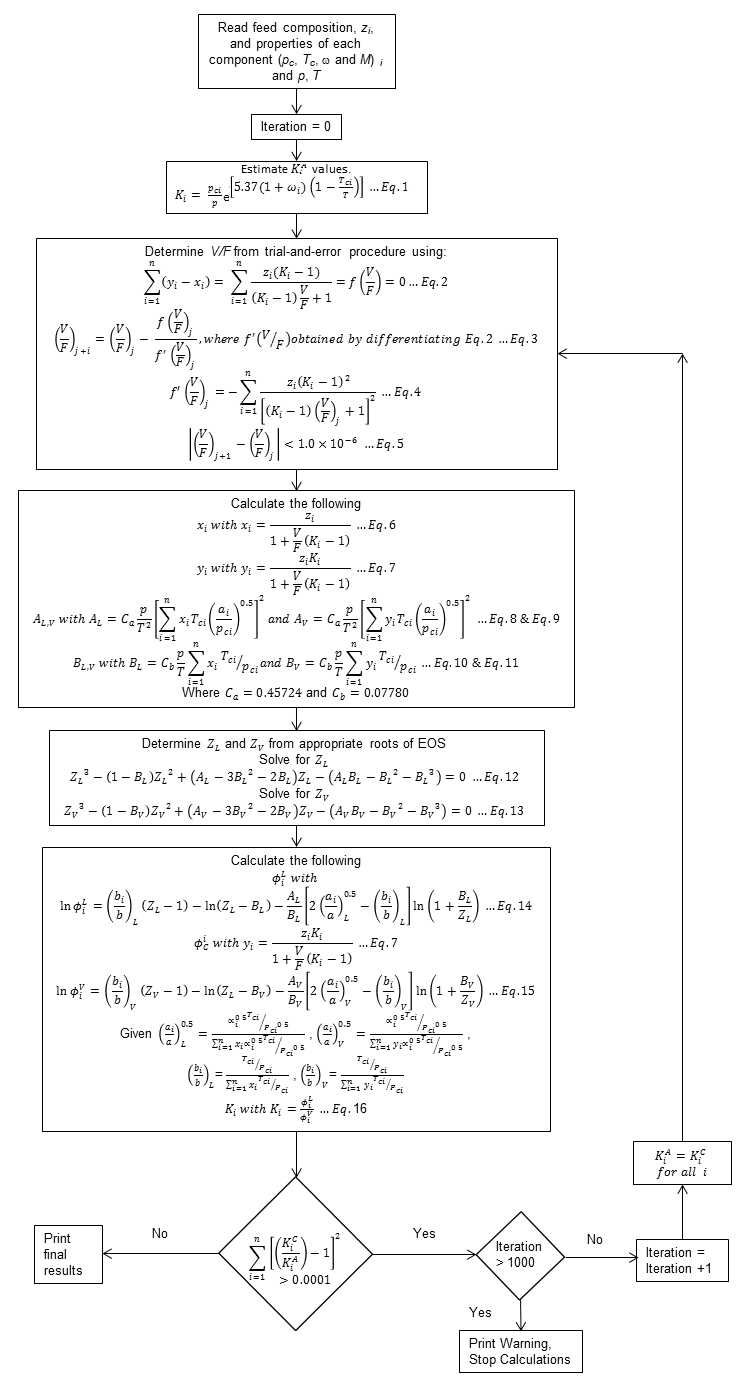
Procedure for calculating gas and oil phases from Equilibrium ratios
Once equilibrium ratios have been solved for, the volumes of each component
Oil Phase:
![]()
Gas Phase:
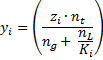
The 12 component composition that Piper uses has one pseudo-component, the C7+fraction.The C7+fraction is intended to represent the combined influence of all components greater than C7in the flash calculation process. Therefore a set of equivalent pseudo-properties need to be calculated for the component. These are described below:
The parameter , which is used in the Standing Equilibrium ratio correlation can be calculated as follows (from PVT and Phase Behavior of Petroleum Fluids - Danesh):
![]()
Where ![]() is calculated as follows (from PVT
and Phase Behavior of Petroleum Fluids - Danesh):
is calculated as follows (from PVT
and Phase Behavior of Petroleum Fluids - Danesh):
![]()
Boiling Point
The boiling point can be calculated from the Soreide correlation (from Phase Behavior – Whitson):
![]()
Critical Properties
Once the boiling point is calculated the critical properties can be calculated for each pseudo-component. The calculation of critical temperature and pressure follows the process described by Twu. The process involves a number of calculations but they aren’t difficult (all of the steps below are from Phase Behavior – Whitson) :
1. Calculate ![]() for
each pseudo-component:
for
each pseudo-component: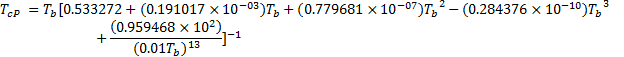
2. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
3. Calculate paraffin specific gravity ![]() for each pseudo-component:
for each pseudo-component:![]()
4. Calculate paraffin critical volume ![]() for each pseudo-component:
for each pseudo-component:![]()
5. Calculate paraffin critical pressure ![]() for each pseudo-component:
for each pseudo-component:![]()
6. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
7. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
8. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
9. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
10. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
11. Calculate ![]() for
each pseudo-component:
for
each pseudo-component: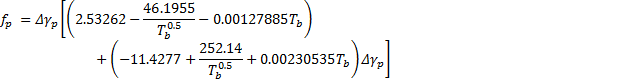
12. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
13. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
14. Calculate ![]() for
each pseudo-component:
for
each pseudo-component:![]()
Acentric Factor
The acentric factor can be calculated from the Lee-Kesler correlations:
1. Calculate the reduced normal boiling point ![]() for the
for the
![]() fraction:
fraction:![]()
2. If ![]() < 0.8, then calculate the acentric
factor
< 0.8, then calculate the acentric
factor ![]() for each pseudo-component using the
following:
for each pseudo-component using the
following:![]()
3. If ![]() 0.8, then calculate
the acentric factor
0.8, then calculate
the acentric factor ![]() for
each pseudo-component using the following:
for
each pseudo-component using the following: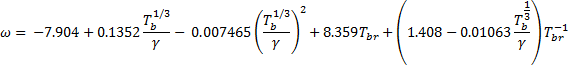
Post-phase calculation properties
Once the phase calculations (either Peng-Robinson or Wilson) has been completed, fluid properties for both the gas and oil phases need to be calculated. This section describes the methods used to calculate each property.
Calculation of Oil Density for Compositional Model
The Standing-Katz method with the Vogel-Yarborough treatment of Nitrogen is used to calculate the density of the oil. Corrections made include nitrogen, ethane density, and methane density.
1. Add the weight fraction of the nitrogen to the weight fraction of the ethane to form a new ethane content for the purpose of this calculation
2. Calculate the molecular weight of the ![]() to
to ![]() with
the following summations:
with
the following summations: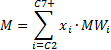
3. Calculate the molar volume of the ![]() to
to ![]() with the following summation, where
(
with the following summation, where
(![]() is
the liquid density of each component:
is
the liquid density of each component: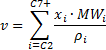
4. Calculate the density of the liquid![]()
5. Calculate the density of the ![]()
![]()
![]()
6. Calculate the molecular weight of the ![]() to
to ![]() components
with the following summations:
components
with the following summations: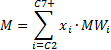
7. Calculate the molar volume of the ![]() to
to ![]() components
with the following summation, where (
components
with the following summation, where (![]() is
the liquid density of each component except for ethane which has its
density replaced with the density calculated in the
is
the liquid density of each component except for ethane which has its
density replaced with the density calculated in the ![]() calculation
above
calculation
above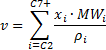
8. Calculate the density of the liquid![]()
9. Calculate the density of the![]()
![]()
![]()
10. Calculate the molecular weight of the ![]() to
to ![]() components
with the following summations:
components
with the following summations: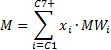
11. Calculate the molar volume of the ![]() to
to ![]() components
with the following summation, where (
components
with the following summation, where (![]() is
the liquid density of each component except for ethane which has its
density replaced with the density calculated in the
is
the liquid density of each component except for ethane which has its
density replaced with the density calculated in the ![]() calculation
above
calculation
above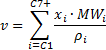
12. Calculate the density of the liquid![]()
The Vogel corrections are applied to Standing-Katz when the in-situ oil density is required.
Calculation of Oil Viscosity for Compositional Model
The Lorenz, Bray and Clark correlation is used to calculate compositional oil viscosity.
1. Calculate the coefficients of the Lorenz, Bray and Clark equation![]()
![]()
![]()
![]()
![]()
2. Calculate the mixture viscosity parameter![]()
3. Calculate the ![]() term
required to calculate reduced oil density
term
required to calculate reduced oil density![]()
4. Calculate the reduced oil density (![]() ):
):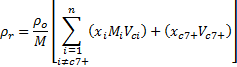
5. Calculate ![]() , which will be
used in the calculation of
, which will be
used in the calculation of ![]()
![]()
6. Calculate ![]() for each component
of Table 2
for each component
of Table 2![]()
If ![]() :
:![]()
If ![]() :
:![]()
7. Calculate the viscosity of the live oil at atmospheric pressure
(![]()
![]()
8. Use the Lorenz, Bray and Clark equation used to calculate viscosity![]()
Gas Compressibility
When Peng-Robinson is selected, the gas compressibility comes directly from the equation of state solution.
When Wilson is selected, gas compressibility is calculated using BWR after converting the full composition into an equivalent gas gravity.
Gas Viscosity
Gas viscosity for both Peng-Robinson and Wilson are calculated using the Carr et al. gas viscosity correlation.
Exclusion of Water from the Compositional Model
Water is not currently considered in the equation of state routine. The exclusion of water was done in the interests of minimizing solution time of the equation of state model, and while keeping in mind that the intent of Piper is to perform pipeline hydraulics calculations, which tend to have only a secondary and tertiary link to the impact of water of condensation and gas dissolved in water.
Instead of including water in the equation of state model black oil like adjustments are made to the gas molar volume and water volume.
Adjustments to dissolved gas in the water are made using the Meehan correlation prior to the equation of state calculation, which therefore effectively removes the gas volume that is dissolved in the water phase.
Adjustments to evaporated water in the gas are made using the Bukacek correlation post-phase volume calculations.
Black Oil in Piper allows for calculation of the change in fluid volumes between in-situ and standard conditions.
When using the Black Oil phase volume calculations, each node in the system will have gas, oil, and water reported in standard conditions and in-situ volumes. While the total standard conditions volumes for each fluid will be consistent throughout the system, the in-situ volumes account for the changing pressure and temperature and the effect on free gas and water relative to those in solution or vapour phase. The image below illustrates the comparison of standard conditions and in-situ volumes.
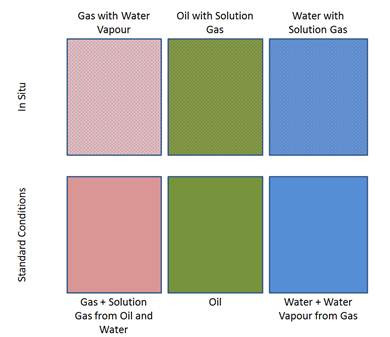
The change in free gas and water for in-situ volumes between two nodes are determined from the Rso, Rsw, and WoC for the pressure and temperature of each node. When the change in pressure and temperature between two nodes result in an increasing Rso or Rsw, there will be less free gas at the in-situ conditions of the second node.
The equations used are listed below. A single equation for each Rsw and WoC are provided. The equation used for Rso is dependent on the correlation selected for Oil PVT in the Correlations dialog.
Solution Gas Oil Ratio
Vasquez & Beggs
Developed from data obtained from fields all over the world and generally applicable for all oil types. Covers a wide range of pressures, temperatures and oil properties.
![]()
![]()
|
Coefficient |
API < 30 |
API > 30 |
|
C1 |
0.0362 |
0.0178 |
|
C2 |
1.0937 |
1.1870 |
|
C3 |
25.7245 |
23.931 |
De Ghetto et al
![]()
![]()
The above equations are suitable for oils with gravity of 10° < API < 22.3°. A modified version of the De Ghetto equations exists for oils below 10°, however this is outside the range of Piper intended use.
Glaso
This correlation is based on North Sea crude-oil data. It is suitable for oil gravity of 22° < API < 48° and believed to be less accurate for Rso > 1400 scf/STBO. This equation cannot be used for p > 19285 psia as a result of the pb* equation.
![]()
![]()
Hanafy et al
This correlation has been developed independent of oil gravity and temperature. Comparisons have demonstrated that it is more applicable to light oils.
![]()
Rso = 0 when p 157.28 psia.
Standing
Applicable for oil gravity of 16° < API < 64°
![]()
Petrosky and Farshad
Applicable for oil gravity of 16° < API < 45°. For Gulf of Mexico oils, it has been found to provide improved results over Standing, Vasquez and Beggs, and Glaso.
![]()
![]()
Velarde at al
Applicable for oil gravity of 12° < API < 55°
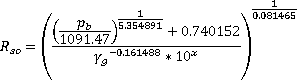
![]()
![]()
Solution Gas Water Ratio
![]()
The above equation describes gas solubility in pure water. The following equation corrects the Rsw for salinity.
![]()
![]()
Water of Condensation
![]()
![]()
![]()
Nomenclature
|
|
Description |
Units |
|
γg |
Gas gravity |
|
|
γAPI |
API oil gravity |
°API |
|
p |
System pressure |
psia |
|
psep |
Separator pressure |
psia |
|
Rso |
Solubility of gas in oil |
scf/STBO |
|
Rsw |
Solubility of gas in water |
scf/STBW |
|
S |
Salinity of water |
Weight % of NaCl |
|
T |
Temperature |
°F |
|
Tsep |
Separator temperature |
°F |
|
WoC |
Water of condensation |
STBW/MMscf |